PASS: Picture Augmentative Synsemic System. Un nuevo sistema para las prácticas habilitativas en la CA (comunicación aumentativa): marco teórico
DOI:
https://doi.org/10.46516/inmaterial.v4.61Palabras clave:
augmentative and alternative communication (AAC), glyph system, habilitation, autism spectrum disorders (ASD), PASSResumen
In this paper we discuss the theoretical linguistic and graphic preconditions of the design of PASS, a glyph system which we designed for use in Augmentative and Alternative Communication (AAC) habilitative practices that has been released under open source licence.
We highlight the relevance of graphic design supporting sustainable practices for people with Autism Spectrum Di- sorders (ASD), in a context in which the o er of public healthcare services for rehabilitation is insufficient.
We present the context in which the AAC is adopted and how a glyph system can be used by people with ASD to learn a language. is particular group of users can access a language by using the glyph system as an interlanguage or as an alternative language.
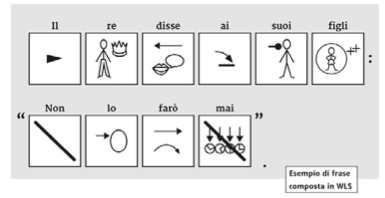
We analyse the most common glyph systems (ARASAAC, PCS, WLS, Blissymbolics), highlighting their strengths and weaknesses from a graphic and linguistic point of view.
We present the theoretical background of the design process for the PASS glyph system.
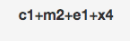
In particular, we provide an in-depth description of the graphic design strategy, which aims to develop a systematic and consistent approach to the construction of the glyphs. This approach is grounded in a reflection on how to solve the linguistic problems raised by the valency model and Chomsky’s generative grammar theory in the visual domain.
We have designed the core of the glyph system by detecting the pertinent visual and linguistic variables in literature, with the objective of developing the system for clinical experimentation.
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2019 Giulia Bonora, Giampiero Dalai, Daniele De Rosa, Marta Panunzi, Luciano Perondi, Cecilia Rubertelli

Esta obra está bajo una licencia internacional Creative Commons Atribución-CompartirIgual 4.0.